Published TCIMAIL newest issue No.199
Maximum quantity allowed is 999
Please select the quantity
CAS RN: 41100-52-1 | Product Number: D3608
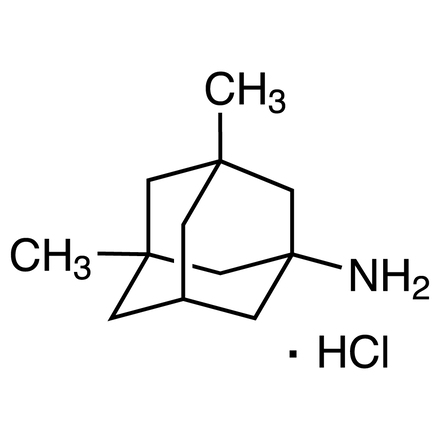
3,5-Dimethyl-1-adamantanamine Hydrochloride
Purity: >98.0%(T)(N)
Synonyms:
- 1-Amino-3,5-dimethyladamantane Hydrochloride
- Memantine Hydrochloride
Product Documents:
| Size | Unit Price | Belgium | Japan* | Quantity |
|---|---|---|---|---|
| 5G |
€58.00
|
1 | 5 |
|
| 25G |
€201.00
|
1 | 17 |
|
*Stock available in Belgium will be delivered in 1 to 3 days
*Stock available in Japan will be delivered in 1 to 2 weeks (excludes regulated items and dry ice shipments).
| Product Number | D3608 |
Purity / Analysis Method 
|
>98.0%(T)(N) |
| Molecular Formula / Molecular Weight | C__1__2H__2__1N·HCl = 215.77 |
| Physical State (20 deg.C) | Solid |
Storage Temperature 
|
Room Temperature (Recommended in a cool and dark place, <15°C) |
| CAS RN | 41100-52-1 |
Related CAS RN 
|
19982-08-2 |
| Reaxys Registry Number | 7937891 |
| PubChem Substance ID | 87559918 |
| Merck Index (14) | 5806 |
| MDL Number | MFCD00214336 |
Specifications
| Appearance | White to Almost white powder to crystal |
| Purity(with Total Nitrogen) | min. 98.0 % |
| Purity(Argentometric Titration) | min. 98.0 % |
Properties (reference)
| Melting Point | 292 °C |
| Solubility in water | Soluble |
GHS
| Pictogram |

|
| Signal Word | Warning |
| Hazard Statements | H315 : Causes skin irritation. H319 : Causes serious eye irritation. |
| Precautionary Statements | P264 : Wash skin thoroughly after handling. P280 : Wear protective gloves/ eye protection/ face protection. P302 + P352 : IF ON SKIN: Wash with plenty of water. P337 + P313 : If eye irritation persists: Get medical advice/ attention. P362 + P364 : Take off contaminated clothing and wash it before reuse. P332 + P313 : If skin irritation occurs: Get medical advice/ attention. |
Related Laws:
| EC Number | 255-219-6 |
Transport Information:
| HS Number | 2921309990 |
Application
Memantine Hydrochloride: A Moderate-Affinity and Uncompetitive N-Methyl-D-Aspartate (NMDA) Receptor Antagonist
Memantine hydrochloride is a moderate-affinity and uncompetitive N-methyl-D-aspartate (NMDA) receptor antagonist. NMDA receptor (NMDAR) is a subtype of glutamate receptor, and very important for controlling synaptic plasticity and memory function. Persistent activation of central nervous system NMDAR by the excitatory amino acid glutamate has been hypothesized to contribute to the symptomatology of Alzheimer’s disease. Memantine binds preferentially in moderate to the NMDAR-operated cation channels, blocks current flow through the NMDARs, and controls the activity of the NMDARs. Therefore, memantine is used for the treatment of Alzheimer’s disease. (The product is for research purpose only.)
References
- Open-channel block of N-methyl-D-aspartate (NMDA) responses by memantine: therapeutic advantage against NMDA receptor-mediated neurotoxicity
- Memantine is a clinically well tolerated N-methyl-D-aspartate (NMDA) receptor antagonist-a review of preclinical data (a review)
- Mechanism of action of Memantine (a review)
- N-methyl D-aspartate (NMDA) receptor antagonists and memantine treatment for Alzheimer's disease, vascular dementia and Parkinson's disease (a review)
- Fluorimetric liquid chromatographic analysis of amantadine in urine and pharmaceutical formulation
- Simultaneous liquid chromatographic assay of amantadine and its four related compounds in phosphate-buffered saline using 4-fluoro-7-nitro-2,1,3-benzoxadiazole [A5593] as a fluorescent derivatization reagent
PubMed Literature
Product Articles
Product Documents (Note: Some products will not have analytical charts available.)
Safety Data Sheet (SDS)
Please select Language.
The requested SDS is not available.
Please Contact Us for more information.
Specifications
C of A & Other Certificates
Please enter Lot Number
Incorrect Lot Number. Please input only the 4-5 alphanumeric characters before the hyphen.
Sample C of A
This is a sample C of A and may not represent a recently manufactured lot of the product.
A sample C of A for this product is not available at this time.
Analytical Charts

Please enter Lot Number
Incorrect Lot Number. Please input only the 4-5 alphanumeric characters before the hyphen.
The requested analytical chart is not available. Sorry for the inconvenience.





