Maximum quantity allowed is 999
CAS RN: 90965-06-3 | Produkte #: D3546
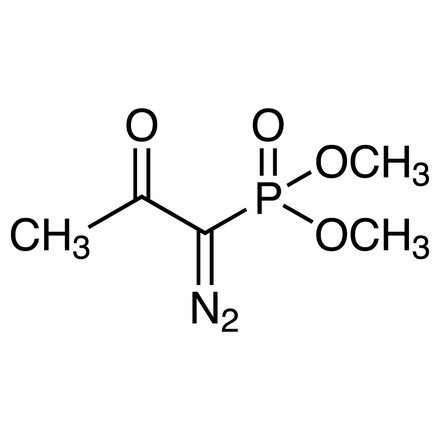
Dimethyl (1-Diazo-2-oxopropyl)phosphonate
Reinheit: >97.0%(HPLC)
- (1-Diazo-2-oxopropyl)phosphonic Acid Dimethyl Ester
- Dimethyl 1-Diazoacetonylphosphonate
- 1-Diazoacetonylphosphonic Acid Dimethyl Ester
- Ohira-Bestmann Reagent
*Lagerbestand in Belgien: Versand am selben Tag
*Lagerbestand in Japan: Bitte prüfen Sie die Versandsimulation für voraussichtliche Lieferzeiten. (ausgenommen regulierte Produkte und Trockeneissendungen)
| Artikel # | D3546 |
Reinheit / Analysenmethode 
|
>97.0%(HPLC) |
| Summenformel / Molekülmasse | C__5H__9N__2O__4P = 192.11 |
| Physikalischer Zustand (20 °C) | Liquid |
Lagerungstemperatur 
|
Refrigerated (0-10°C) |
| Unter Inertgas lagern | Store under inert gas |
| Zu vermeidende Bedingungen | Air Sensitive,Heat Sensitive |
| CAS RN | 90965-06-3 |
| Reaxys Registrierungsnummer | 4247670 |
| PubChem-Stoff-ID | 87559944 |
| Merck-Index (14) | 1177 |
| MDL-Nummer | MFCD07368360 |
| Appearance | Light yellow to Brown clear liquid |
| Purity(HPLC) | min. 97.0 area% |
| Spezifisches Gewicht (20/20) | 1.28 |
| Brechungsindex | 1.48 |
| HS-Nr. (Import / Export) (TCI-E) | 2931498090 |

-
Used Chemicals
-
Procedure
-
To a methanol (10 mL) solution of undecanal (0.20 g, 1.2 mmol) was added potassium carbonate (0.32 g,2.4 mmol) and Ohira-Bestmann reagent (0.27 g, 1.4 mmol) at room temperature. The reaction mixture was stirred overnight at room temperature, then diluted with diethyl ether and washed with saturated aqueous sodium bicarbonate, and dried by sodium sulfate. The organic layer was concentrated under reduced pressure. The resulting crude product was purified by column chromatography (hexane:toluene = 3:1 on silica gel) to give 1 as a colorless liquid (0.11 g, 56% yield).
-
Experimenter’s Comments
-
The reaction mixtures were monitored by 1H NMR (CDCl3).
-
Analytical Data(1-Dodecyne (1))
-
1H NMR (400 MHz, CDCl3); δ 2.18 (t, 2H, J = 7.1 Hz), 1.94 (s, 1H), 1.57-1.46 (m, 2H), 1.44-1.33 (m, 2H), 1.33-1.19 (m, 12H), 0.88 (t, 3H, J = 6.4 Hz).
13C NMR (101 MHz, CDCl3); δ 85.0, 68.2, 32.0, 29.7, 29.7, 29.5, 29.3, 28.9, 28.6, 22.8, 18.5, 14.3.
-
Lead Reference
-
- Further Improvements of the Synthesis of Alkynes from Aldehydes
-
Other References
-
- Methanolysis of Dimethyl (1-Diazo-2-oxopropyl) Phosphonate: Generation of Dimethyl (Diazomethyl) Phosphonate and Reaction with Carbonyl Compounds

References

References
- S. Ohira, Synth. Commun. 1989, 19, 561.

- S. Muller, B. Liepold, G. J. Roth, H. J. Bestmann, Synlett 1996, 521.

- H. D. Dickson, S. C. Smith, K. W. Hinkle, Tetrahedron Lett. 2004, 45, 5597.

- E. Quesada, R. J. K. Taylor, Tetrahedron Lett. 2005, 46, 6473.

Dokumente
[TCI Practical Example] Construction of the Terminal Alkyne Using Ohira-Bestmann Reagent
Sicherheitsdatenblatt (SDB)
Das angeforderte SDB ist nicht verfügbar.
Bitte Kontaktieren Sie uns für mehr Informationen.
Spezifikationsdokumenten
AZ & andere Zertifikate
Muster-AZ
Ein Muster-AZ für dieses Produkt ist zur Zeit nicht verfügbar.
Analytische Diagramme

Das angeforderte Analysediagramm ist nicht verfügbar. Wir entschuldigen uns für die Unannehmlichkeiten.