Published TCIMAIL newest issue No.199
Maximum quantity allowed is 999
CAS RN: 153254-09-2 | Product Number: B3022
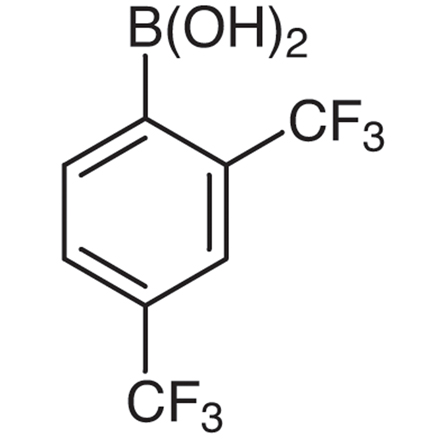
2,4-Bis(trifluoromethyl)phenylboronic Acid (contains varying amounts of Anhydride)
Purity:
- 2,4-Bis(trifluoromethyl)benzeneboronic Acid (contains varying amounts of Anhydride)
| Size | Unit Price | Belgium | Japan* | Quantity |
|---|---|---|---|---|
| 1G |
£35.00
|
Contact Us | 1 |
|
| 5G |
£114.00
|
1 | 30 |
|
*Stock available in Belgium will be delivered in 1 to 3 days
*Stock available in Japan will be delivered in 1 to 2 weeks (excludes regulated items and dry ice shipments).
| Product Number | B3022 |
| Molecular Formula / Molecular Weight | C__8H__5BF__6O__2 = 257.93 |
| Physical State (20 deg.C) | Solid |
Storage Temperature 
|
Room Temperature (Recommended in a cool and dark place, <15°C) |
| CAS RN | 153254-09-2 |
| Reaxys Registry Number | 10175108 |
| PubChem Substance ID | 87559527 |
| MDL Number | MFCD01631349 |
| Appearance | White to Light yellow powder to crystal |
| Purity(Neutralization titration) | 97.0 to 108.0 % |
| NMR | confirm to structure |
| Solubility (soluble in) | Methanol |
| Pictogram |

|
| Signal Word | Warning |
| Hazard Statements | H315 : Causes skin irritation. H319 : Causes serious eye irritation. |
| Precautionary Statements | P264 : Wash skin thoroughly after handling. P280 : Wear protective gloves/ eye protection/ face protection. P302 + P352 : IF ON SKIN: Wash with plenty of water. P337 + P313 : If eye irritation persists: Get medical advice/ attention. P362 + P364 : Take off contaminated clothing and wash it before reuse. P332 + P313 : If skin irritation occurs: Get medical advice/ attention. |
| HS Number | 2931900090 |
Reference
- Inhibition of Quorum Sensing in Vibrio harveyi by Boronic Acids
-
Used Chemicals
-
Procedure
-
To a solution of benzoic acid (122 mg, 1.0 mmol) and benzylamine (107 mg, 1.0 mmol) in fluorobenzene (5 mL, 0.2 mol/L) was added molecular sieves 3A (2 g) at room temperature. After that 2,4-bis(trifluoromethyl)phenylboronic acid (13 mg, 0.05 mmol) was added to the reaction mixture and stirred at reflux temperature for 24 hours. The reaction mixture was filtered on celite pad and washed with EtOAc. The filtrate was washed with 2 mol/L HCl aq. (30 mL), saturated NaHCO3 aq. (30 mL), and brine (30 mL), dried over sodium sulfate and filtered. The solvent was removed under reduced pressure to give N-benzylbenzamide as a white solid (135 mg, 64%).
-
Experimenter’s Comments
-
The reaction mixture was monitored by LC.
-
Analytical Data
-
N-benzylbenzamide
1H NMR (270 MHz, DMSO-d6); δ 9.05 (brt, 1H, J = 5.4 Hz), 7.90 (dd, 1H, J = 7.3, 1.6 Hz), 7.56-7.44 (m, 3H), 7.36-7.32 (m, 4H), 7.25 (q, 1H, J = 7.3 Hz).
-
Lead Reference
-
- The ortho-substituent on 2,4-bis(trifluoromethyl)phenylboronic acid catalyzed dehydrative condensation between carboxylic acids and amines

Safety Data Sheet (SDS)
The requested SDS is not available.
Please Contact Us for more information.
Specifications
C of A & Other Certificates
Sample C of A
A sample C of A for this product is not available at this time.
Analytical Charts

The requested analytical chart is not available. Sorry for the inconvenience.





