Published TCIMAIL newest issue No.199
Maximum quantity allowed is 999
CAS RN: 16940-66-2 | Product Number: S0480
Sodium Borohydride

Purity: >95.0%(T)
- Sodium Tetrahydroborate
- Sodium Tetrahydridoborate
| Size | Unit Price | Belgium | Japan* | Quantity |
|---|---|---|---|---|
| 25G |
£21.00
|
22 | ≥60 |
|
| 100G |
£39.00
|
25 | ≥60 |
|
| 500G |
£103.00
|
≥80 | ≥60 |
|
*Stock available in Belgium will be delivered in 1 to 3 days
*Stock available in Japan will be delivered in 1 to 2 weeks (excludes regulated items and dry ice shipments).
| Product Number | S0480 |
Purity / Analysis Method 
|
>95.0%(T) |
| Molecular Formula / Molecular Weight | NaBH__4 = 37.83 |
| Physical State (20 deg.C) | Solid |
Storage Temperature 
|
Room Temperature (Recommended in a cool and dark place, <15°C) |
| Store Under Inert Gas | Store under inert gas |
| Condition to Avoid | Moisture Sensitive |
| CAS RN | 16940-66-2 |
| Reaxys Registry Number | 11343078 |
| PubChem Substance ID | 87575954 |
| Merck Index (14) | 8592 |
| MDL Number | MFCD00003518 |
| Appearance | White to Almost white powder to crystal |
| Purity(Sodium hypochlorite Method) | min. 95.0 % |
| Solubility in water | Soluble |
| Solubility (insoluble in) | Ether |
| Pictogram |




|
| Signal Word | Danger |
| Hazard Statements | H301 + H311 : Toxic if swallowed or in contact with skin. H314 : Causes severe skin burns and eye damage. H360 : May damage fertility or the unborn child. H260 : In contact with water releases flammable gases which may ignite spontaneously. |
| Precautionary Statements | P260 : Do not breathe dust. P231 + P232 : Handle and store contents under inert gas. Protect from moisture. P201 : Obtain special instructions before use. P280 : Wear protective gloves/ protective clothing/ eye protection/ face protection/ hearing protection. P370 + P378 : In case of fire: Use dry sand, dry chemical or alcohol-resistant foam to extinguish. P308 + P313 : IF exposed or concerned: Get medical advice/ attention. P303 + P361 + P353 : IF ON SKIN (or hair): Take off immediately all contaminated clothing. Rinse skin with water. P302 + P335 + P334 : IF ON SKIN: Brush off loose particles from skin. Immerse in cool water. P301 + P310 + P330 : IF SWALLOWED: Immediately call a POISON CENTER/doctor. Rinse mouth. P304 + P340 + P310 : IF INHALED: Remove person to fresh air and keep comfortable for breathing. Immediately call a POISON CENTER/doctor. P305 + P351 + P338 + P310 : IF IN EYES: Rinse cautiously with water for several minutes. Remove contact lenses, if present and easy to do. Continue rinsing. Immediately call a POISON CENTER/doctor. |
| EC Number | 241-004-4 |
| RTECS# | ED3325000 |
| UN Number | UN1426 |
| Class | 4.3 |
| Packing Group | I |
| HS Number | 2850002090 |

-
Used Chemicals
-
Procedure
-
Sodium borohydride (250 mg, 6.70 mmol) was separately added to a solution of 4',5'-dimethoxy-2'-nitroacetophenone (1.50 g, 6.70 mmol) in THF (12 mL) and methanol (3 mL) at 0 °C. Then the mixture was allowed to warm to room temperature and stirred for 1.5 hours. The solution was diluted with dichloromethane (15 mL) and washed with water (15 mL x 3) and brine (15 mL), dried over sodium sulfate and filtered. The solvent was removed under reduced pressure. The residue was diluted with THF (6 mL) and the mixture was stirred at 37 °C. Hexane (10 mL) was added to the solution and the mixture was stirred at 0 °C and filtered to give 1 (1.16 g、77%) as a yellow solid.
-
Experimenter’s Comments
-
The reaction mixture was monitored by TLC (hexane:ethyl acetate = 1:1, Rf = 0.47) and UPLC.
To avoid moisture absorption of sodium borohydride, weighing was done quickly.
-
Analytical Data
-
Compound 1
1H NMR (400 MHz, CDCl3); δ 7.57 (s, 1H), 7.31 (s, 1H), 5.58 (q, J = 5.4 Hz, 1H), 4.01 (s, 3H), 3.95 (s, 3H), 1.56 (d, J = 5.4 Hz, 3H).
-
Lead Reference
-
- Triggering a [2]Rotaxane Molecular Shuttle by a Photochemical Bond-Cleavage Strategy
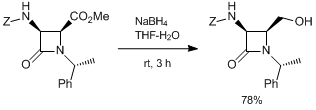
References
- Stereoselective synthesis of .beta.-lactams by oxidative coupling of dianions of acyclic tertiary amides
- Forty years of hydride reductions
- Reductions by the alumino- and borohydrides in organic synthesis

References
- An ultrasafe hydrogen generator: aqueous, alkaline borohydride solutions and Ru catalyst.
- A Compact borohydride fuel cell.
- A fuel cell development for using borohydrides as the fuel
- A study on hydrogen generation from NaBH4 solution using the high-performance Co-B catalyst
- A comprehensive review of direct borohydride fuel cells
- Accelerating velocity of hydrogen generation from sodium borohydride by citric acid catalyst

References
Documents
[Research Articles] Nickel Boride Mediated Cleavage of 1,3-Dithiolanes
Safety Data Sheet (SDS)
The requested SDS is not available.
Please Contact Us for more information.
Specifications
C of A & Other Certificates
Sample C of A
A sample C of A for this product is not available at this time.
Analytical Charts

The requested analytical chart is not available. Sorry for the inconvenience.






