Published TCIMAIL newest issue No.199
Maximum quantity allowed is 999
Please select the quantity
CAS RN: 7562-61-0 | Product Number: U0023
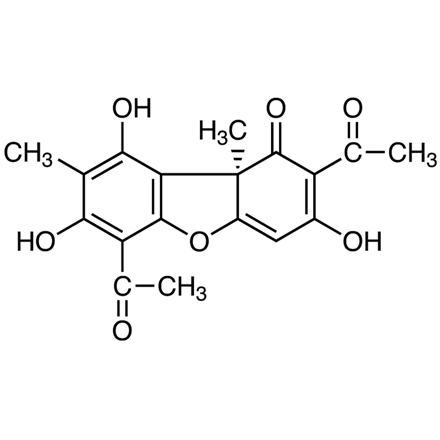
(+)-Usnic Acid
Purity: >98.0%(T)(HPLC)
Synonyms:
Product Documents:
| Size | Unit Price | Belgium | Japan* | Quantity |
|---|---|---|---|---|
| 1G |
£16.00
|
1 | 9 |
|
| 25G |
£155.00
|
Contact Us | 9 |
|
*Stock available in Belgium will be delivered in 1 to 3 days
*Stock available in Japan will be delivered in 1 to 2 weeks (excludes regulated items and dry ice shipments).
| Product Number | U0023 |
Purity / Analysis Method 
|
>98.0%(T)(HPLC) |
| Molecular Formula / Molecular Weight | C__1__8H__1__6O__7 = 344.32 |
| Physical State (20 deg.C) | Solid |
Storage Temperature 
|
Room Temperature (Recommended in a cool and dark place, <15°C) |
Packaging and Container 
|
1G-Glass Bottle with Plastic Insert (View image) |
| CAS RN | 7562-61-0 |
| Reaxys Registry Number | 96698 |
| PubChem Substance ID | 87577753 |
| SDBS (AIST Spectral DB) | 7871 |
| Merck Index (14) | 9893 |
| MDL Number | MFCD00016878 |
Specifications
| Appearance | Light orange to Yellow to Green powder to crystal |
| Purity(HPLC) | min. 98.0 area% |
| Purity(Neutralization titration) | min. 98.0 % |
| Melting point | 195.0 to 203.0 °C |
| Specific rotation [a]20/D | +485 to +505 deg(C=0.7, CHCl3) |
| Solubility in Chloroform | almost transparency |
| NMR | confirm to structure |
Properties (reference)
| Melting Point | 200 °C |
| Specific Rotation | 495° (C=0.7,CHCl3) |
GHS
| Pictogram |


|
| Signal Word | Danger |
| Hazard Statements | H302 : Harmful if swallowed. H370 : Causes damage to organs. |
| Precautionary Statements | P501 : Dispose of contents/ container to an approved waste disposal plant. P260 : Do not breathe dust. P270 : Do not eat, drink or smoke when using this product. P264 : Wash skin thoroughly after handling. P308 + P311 : IF exposed or concerned: Call a POISON CENTER/doctor. P301 + P312 + P330 : IF SWALLOWED: Call a POISON CENTER/doctor if you feel unwell. Rinse mouth. |
Related Laws:
| EC Number | 231-456-0 |
| RTECS# | HP5295050 |
Transport Information:
| HS Number | 2932990090 |
Application
(+)-Usnic acid: A Lichen Metabolite with Antibiotic and Several Interesting Properties

(+)-Usnic acid is one of the most common and abundant lichen metabolites, and well known its antibiotic and several interesting properties.1,2) It is reported that the (+)-usnic acid is effective against a large variety Gram-positive bacterial stains including multi-resistant stains of Streptococcus aureus, enterococci and mycobacteria. (+)-Usnic acid also appears to be against Streptococcus mutans.
On the chemical structure, the absolute configuration of (+)-usnic acid at 9b position has determined by X-ray analysis to be R. 2,3,4) Of plausible tautomers, the low energy 1-oxo, 3-hydroxy confirmation is preferred according to calculations.1,2,3) The enolic 3-hydroxy group has the strongest acidic character (pKa 4.4) due to an inductive effect of the keto group.2) Recently, (+)-usnic acid derivatives are used as an organic ligand.5)
On the chemical structure, the absolute configuration of (+)-usnic acid at 9b position has determined by X-ray analysis to be R. 2,3,4) Of plausible tautomers, the low energy 1-oxo, 3-hydroxy confirmation is preferred according to calculations.1,2,3) The enolic 3-hydroxy group has the strongest acidic character (pKa 4.4) due to an inductive effect of the keto group.2) Recently, (+)-usnic acid derivatives are used as an organic ligand.5)
References
- Study of the cytotoxic and antimicrobial activities of usnic acid and derivatives
- E. R. Correche, M. Carrasco, M. E. Escudero, L. Velazquez, A. M. S. De Guzman, F. Giannini, R. D. Enriz, E. A. Jauregui, J. P. Cenal, O. S. Giordano, Fitoterapia 1998, 69, 493.
- Molecules of Interest usnic acid (a review)
- New Results on the Chemistry of Lichen Substances, 4.5.13. Usnic Acid and Related Compounds
- The absolute configurations of (+)-usnic and (+)-isousnic acid. X-ray analyses of the (-)-α-phenylethylamine derivative of (+)-usnic acid and of (-)-pseudoplacodiolic acid, a new dibenzofuran, from the lichen Rhizoplaca chrysoleuca.
- Novel chiral molecular tweezer from (+)-usnic acid
PubMed Literature
Documents
Product Articles
Product Documents (Note: Some products will not have analytical charts available.)
Safety Data Sheet (SDS)
Please select Language.
The requested SDS is not available.
Please Contact Us for more information.
Specifications
C of A & Other Certificates
Please enter Lot Number
Incorrect Lot Number. Please input only the 4-5 alphanumeric characters before the hyphen.
Sample C of A
This is a sample C of A and may not represent a recently manufactured lot of the product.
A sample C of A for this product is not available at this time.
Analytical Charts

Please enter Lot Number
Incorrect Lot Number. Please input only the 4-5 alphanumeric characters before the hyphen.
The requested analytical chart is not available. Sorry for the inconvenience.





