Published TCIMAIL newest issue No.199
Maximum quantity allowed is 999
* Stock available in Belgium: Shipment on the same day
* Stock available in Japan: Please check the Shipping Simulation for estimated shipments. (excludes regulated items and dry ice shipments)
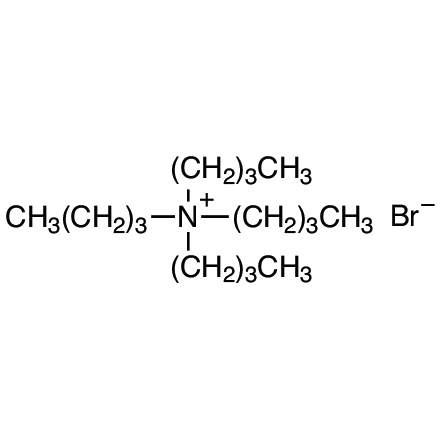
| Product Number | T0054 |
Purity / Analysis Method 
|
>98.0%(T) |
| Molecular Formula / Molecular Weight | C__1__6H__3__6BrN = 322.38 |
| Physical State (20 deg.C) | Solid |
Storage Temperature 
|
Room Temperature (Recommended in a cool and dark place, <15°C) |
| Store Under Inert Gas | Store under inert gas |
| Condition to Avoid | Hygroscopic |
| CAS RN | 1643-19-2 |
| Reaxys Registry Number | 3570983 |
| PubChem Substance ID | 87576073 |
| SDBS (AIST Spectral DB) | 3131 |
| MDL Number | MFCD00011633 |
| Appearance | White to Almost white powder to crystaline |
| Purity(Nonaqueous Titration) | min. 98.0 % |
| Drying loss | max. 1.0 % |
| pH | 3.5 to 7.0 (50 g/L, 25 deg-C) |
| Solubility in Water | almost transparency |
| Tributylamine hydrobromide | max. 1.0 % |
| Melting Point | 103 °C |
| Solubility in water | Soluble |
| Solubility (soluble in) | Alcohol |
| Pictogram |

|
| Signal Word | Warning |
| Hazard Statements | H302 : Harmful if swallowed. H315 : Causes skin irritation. H319 : Causes serious eye irritation. |
| Precautionary Statements | P501 : Dispose of contents/ container to an approved waste disposal plant. P270 : Do not eat, drink or smoke when using this product. P264 : Wash skin thoroughly after handling. P280 : Wear protective gloves/ eye protection/ face protection. P337 + P313 : If eye irritation persists: Get medical advice/ attention. P301 + P312 + P330 : IF SWALLOWED: Call a POISON CENTER/doctor if you feel unwell. Rinse mouth. |
| EC Number | 216-699-2 |
| HS Number | 2923900055 |




The requested SDS is not available.
Please Contact Us for more information.
A sample C of A for this product is not available at this time.

The requested analytical chart is not available. Sorry for the inconvenience.