Published TCIMAIL newest issue No.200
Maximum quantity allowed is 999
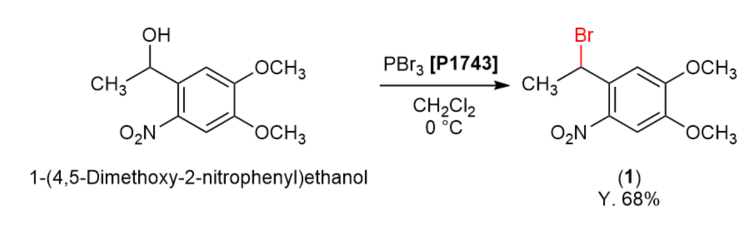
TCI Practical Example: Bromination Using Phosphorus Tribromide
We are proud to present the bromination of 1-(4,5-dimethoxy-2-nitrophenyl)ethanol using phosphorus tribromide.
Used Chemicals
- Phosphorus Tribromide [P1743]
- 1-(4,5-Dimethoxy-2-nitrophenyl)ethanol
- Dichloromethane
Procedure
Phosphorus tribromide (1.63 g, 6.03 mmol) was added dropwise to a solution of 1-(4,5-dimethoxy-2-nitrophenyl)ethanol (1.10 g, 4.83 mmol) in dichloromethane (24 mL) at 0 °C and the mixture was stirred for 3 hours. Then the reaction was quenched with saturated sodium hydrogen carbonate aqueous solution (18 mL) and the two layers were partitioned. The organic layer was washed with water (10 mL, 2 times), dried over sodium sulfate and filtered. The solvent was removed under reduced pressure and the residue was purified by column chromatography (on silica gel, ethyl acetate:hexane = 1:10) to give compound 1 (952 mg, 68% yield) as a yellow oil.
Experimenter's Comments
The reaction mixture was monitored by TLC (hexane:ethyl acetate = 1:1, Rf = 0.71) and UPLC.
Compound 1 was treated under light shielding throughout the entire process because of its possible photodegradability.
When the sample for NMR of 1 was left under fluorescent light, the color of the solution changed from yellow to dark blue.Analytical Data
Compound 1
1H NMR (400 MHz, CDCl3); δ 7.46 (s, 1H), 7.28 (s, 1H), 6.04 (q, J = 5.4 Hz, 1H), 4.02 (s, 3H), 3.95 (s, 3H), 2.07 (d, J = 5.4 Hz, 3H).
Lead Reference
- Photolabile Protecting Groups for Nitroxide Spin Labels

