Maintenance Notice (3:30 AM August 30 - 12:00 PM August 31, 2025 UTC): This website is scheduled to be unavailable due to maintenance.
During the period, there is case that you can access this website, but you would face errors. Please wait until notice of the maintenance completion. We appreciate your patience and understanding.
Maximum quantity allowed is 999
Please select the quantity
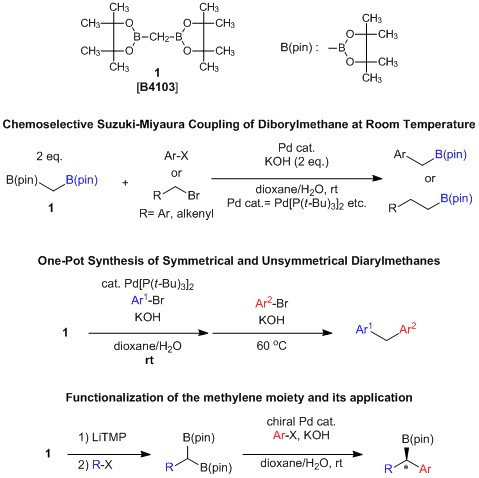
A Useful Methylene Unit Usable for Suzuki-Miyaura Cross-coupling
Bis(pinacolatoboryl)methane (1) consists of a methylene group and two pinacolatoboryl groups, and can be used for introducing two different functional groups continuously by the use of a palladium catalyst. For example, in the presence of bis(tri-tert-butylphosphine)palladium(0) and potassium hydroxide, 1 reacts with aryl halides at room temperature to afford the desired benzylboronates selectively. In this reaction, sp3-carbon and sp3-carbon forming reactions successfully proceed by use of allyl bromides and benzyl bromides.1-3) Also, when 1 reacts with aryl halides in two parts, symmetrical diarylmethanes are given. If two different aryl halides react with 1 in order, asymmetrical diarylmethanes are synthesized in one pot.4) Furthermore, deprotonation of a methylene proton of 1 can occur by the treatment of LiTMP, and a consequent reaction with alkyl halides affords alkylated diborylmethanes. The given products are applied to the asymmetric Suzuki-Miyaura cross-coupling by use of chiral ligands.5)
Related Products
References
- K. Endo, T. Ohkubo, T. Shibata, Org. Lett. 2011, 13, 3368.

- K. Endo, T. Ohkubo, T. Ishioka, T. Shibata, J. Org. Chem. 2012, 77, 4826.

- K. Endo, T. Ishioka, T. Shibata, Synlett 2014, 25, 2184.

- K. Endo, T. Ishioka, T. Ohkubo, T. Shibata, J. Org. Chem. 2012, 77, 7223.

- C. Sun, B. Potter, J. P. Morken, J. Am. Chem. Soc. 2014, 136, 6534.
