Maintenance notice in CEST time zone (1:00 - 3:30 AM May 17, 2026): Our website is scheduled for maintenance. The website will be available, but some functions may experience errors. We appreciate your patience and understanding.
Published TCIMAIL newest issue No.201
Maximum quantity allowed is 999
Please select the quantity
CAS RN: 929896-28-6 | Product Number: B4195
Bis(acetato)aqua[(S,S)-4,6-bis(4-isopropyl-2-oxazolin-2-yl)-m-xylene]rhodium
Purity:
Synonyms:
Product Documents:
| Size | Unit Price | Belgium | Japan* | Quantity |
Shipping Information

|
|---|---|---|---|---|---|
| 10MG |
143,00 €
|
1 | Contact Us |
|
* Stock available in Belgium: Shipment on the same day
* Stock available in Japan: Please check the Shipping Simulation for estimated shipments. (excludes regulated items and dry ice shipments)
| Product Number | B4195 |
| Molecular Formula / Molecular Weight | C__2__4H__3__5N__2O__7Rh = 566.46 |
| Physical State (20 deg.C) | Solid |
Storage Temperature 
|
Room Temperature (Recommended in a cool and dark place, <15°C) |
| CAS RN | 929896-28-6 |
| PubChem Substance ID | 253659893 |
Specifications
| Appearance | Light yellow to Yellow to Orange powder to crystal |
| Elemental analysis(Nitrogen) | 4.40 to 5.20 % |
| NMR | confirm to structure |
Properties (reference)
GHS
Related Laws:
Transport Information:
| HS Number | 2843909000 |
Application
Pincer Type Rh Complex Catalyzed Asymmetric Diboration of Terminal Alkenes and Their Conversion to the Optically Active 1,2-Diols
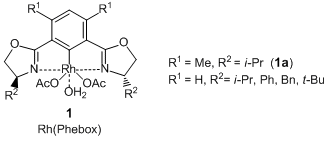

Nishiyama et al. have reported that optically active 1,2-diols are given by the pincer type Rh complexes [Rh(Phebox), 1] catalyzed asymmetric 1,2-diboration of terminal alkenes and subsequent oxidation. According to their results, the asymmetric diboration of 4-chlorostyrene with a diboron ester proceeds with high enantioselectivity by using a rhodium complex coordinated with a pincer type ligand (1a), and the desired 1,2-diborylated product is formed. Subsequent oxidation of it with sodium peroxoborate in THF-water gives the corresponding 1,2-diol. In this reaction, allyl ethers and allyl anilines can be transformed into the related optically active 1,2-diols. In addition, when using trans 1,3-dienes as reactants, terminal alkene moieties are selectively reacted and the optically active allylic alcohols are given. Thus, this asymmetric diboration using 1 is an efficient synthetic method to produce the optically active 1,2-diols.
References
- Asymmetric diboration of terminal alkenes with a rhodium catalyst and subsequent oxidation: enantioselective synthesis of optically active 1,2-diols
PubMed Literature
Product Articles
[TCIMAIL No.160] Pincer Type Rh Complex Catalyzed Asymmetric Diboration of Terminal Alkenes and Their Conversion to the Optically Active 1,2-DiolsProduct Documents (Note: Some products will not have analytical charts available.)
Safety Data Sheet (SDS)
Please select Language.
The requested SDS is not available.
Please Contact Us for more information.
Specifications
C of A & Other Certificates
Please enter Lot Number
Incorrect Lot Number. Please input only the 4-5 alphanumeric characters before the hyphen.
Sample C of A
This is a sample C of A and may not represent a recently manufactured lot of the product.
A sample C of A for this product is not available at this time.
Analytical Charts

Please enter Lot Number
Incorrect Lot Number. Please input only the 4-5 alphanumeric characters before the hyphen.
The requested analytical chart is not available. Sorry for the inconvenience.


![Bis(acetato)aqua[(S,S)-4,6-bis(4-isopropyl-2-oxazolin-2-yl)-m-xylene]rhodium Thumbnail of Bis(acetato)aqua[(S,S)-4,6-bis(4-isopropyl-2-oxazolin-2-yl)-m-xylene]rhodium](/medias/B4195.jpg?context=bWFzdGVyfHJvb3R8MTc3NDB8aW1hZ2UvanBlZ3xhR0UxTDJobU1pODRPVE16TmpjeU1Ea3hOamM0TDBJME1UazFMbXB3Wnd8YjUzYTM1YjZlZGNhYmU3OGMxM2EyOTkyNWQyYWZjNzc3NDBhMDEwZGZkZmFmOTNhOTVjOWY0NjQzY2YzMmY5MA)
![Bis(acetato)aqua[(S,S)-4,6-bis(4-isopropyl-2-oxazolin-2-yl)-m-xylene]rhodium Chemical Structure of Bis(acetato)aqua[(S,S)-4,6-bis(4-isopropyl-2-oxazolin-2-yl)-m-xylene]rhodium](/medias/B4195.jpg?context=bWFzdGVyfHJvb3R8NjUyNzd8aW1hZ2UvanBlZ3xhRGsxTDJnMFlpODRPVEl3TWpVNE1UZ3pNVGs0TDBJME1UazFMbXB3Wnd8NTM3ZDVkNGU3NWI1NjY5Y2VhNjM2YzAzODdmM2U3MjhhOGVhNjM1ZGM0MTExNGM2MWJmNjgyYjE5NWFjM2YyNQ)
![Bis(acetato)aqua[(S,S)-4,6-bis(4-isopropyl-2-oxazolin-2-yl)-m-xylene]rhodium Bis(acetato)aqua[(S,S)-4,6-bis(4-isopropyl-2-oxazolin-2-yl)-m-xylene]rhodium](/_ui/responsive/theme-tci/images/missing_product_zoom.webp)

