Maximum quantity allowed is 999
CAS RN: 131274-22-1 | 产品编码: T2584
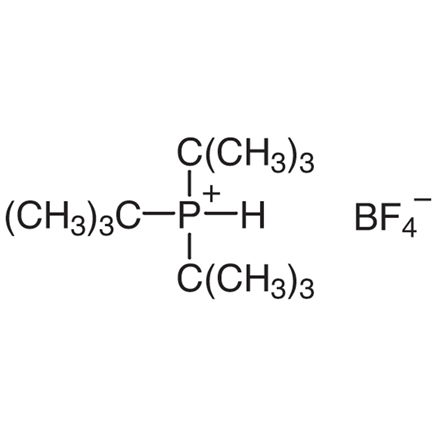
Tri-tert-butylphosphonium Tetrafluoroborate
| 产品编码 | T2584 |
纯度/分析方法 
|
>98.0%(T) |
| 分子式/分子量 | C__1__2H__2__8BF__4P = 290.13 |
| 外观与形状(20°C) | 固体 |
储存温度 
|
室温 (15°C以下阴凉干燥处) |
包装和容器 
|
1G-Glass Bottle with Plastic Insert (查看图片) |
| CAS RN | 131274-22-1 |
| Reaxys-RN | 8813613 |
| PubChem物质ID | 125307337 |
| MDL编号 | MFCD04039975 |
| Appearance | White to Almost white powder to crystal |
| Purity(Iodometric Titration) | min. 98.0 % |
| NMR | confirm to structure |
| 熔点 | 261 °C |
| 象形图 |


|
| 信号词 | Danger |
| 危险性说明 | H302 : Harmful if swallowed. H314 : Causes severe skin burns and eye damage. |
| 防范说明 | P501 : Dispose of contents/ container to an approved waste disposal plant. P260 : Do not breathe dust. P270 : Do not eat, drink or smoke when using this product. P264 : Wash skin thoroughly after handling. P280 : Wear protective gloves/ protective clothing/ eye protection/ face protection. P303 + P361 + P353 : IF ON SKIN (or hair): Take off immediately all contaminated clothing. Rinse skin with water. P301 + P330 + P331 : IF SWALLOWED: Rinse mouth. Do NOT induce vomiting. P363 : Wash contaminated clothing before reuse. P301 + P312 + P330 : IF SWALLOWED: Call a POISON CENTER/doctor if you feel unwell. Rinse mouth. P304 + P340 + P310 : IF INHALED: Remove person to fresh air and keep comfortable for breathing. Immediately call a POISON CENTER/doctor. P305 + P351 + P338 + P310 : IF IN EYES: Rinse cautiously with water for several minutes. Remove contact lenses, if present and easy to do. Continue rinsing. Immediately call a POISON CENTER/doctor. P405 : Store locked up. |
| UN编号 | UN1759 |
| 类别 | 8 |
| 包装类别 | III |
| HS编码* | 2931.49-900 |
Used Chemicals
Procedure
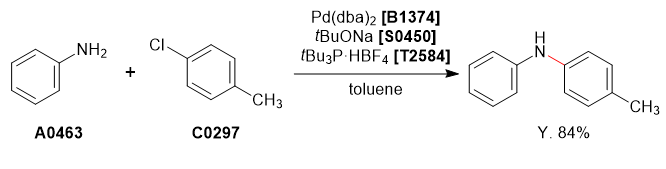
Aniline (1.0 g, 10.7 mmol) and 4-chlorotoluene (1.4 g, 11.8 mmol) were dissolved in degassed toluene (10 mL) under nitrogen atmosphere. To this solution was added sodium tert-butoxide (1.54 g, 16.1 mmol), tri-tert-butylphosphonium tetrafluoroborate (31.1 mg, 0.1 mmol) and Pd(dba)2 (61.7 mg, 0.1 mmol, 1.5mol%). The reaction mixture was refluxed for 24 h and cooled to room temperature and quenched with H2O (10 mL). The organic phase was separated and washed with brine (10 mL), dried over MgSO4 (10 g), concentrated under reduced pressure to afford the crude product as a brown liquid. The crude product was purified by column chromatography on silica-gel (hexane : ethyl acetate = 95 : 5) to afford the desired product as a pale yellow crystalline solid (1.21 g, 6.6 mmol, 62%).
Experimenter’s Comments
Toluene was degassed by bubbling with N2 gas for 30 min.
The reaction mixture was analyzed by TLC (hexane : ethyl acetate = 1 : 1, Rf = 0.30) and GC.
Analytical Data (4-Methyl-N-phenylaniline)
1H NMR (400 MHz, CDCl3); δ 7.24-7.20 (t, J = 8.7 Hz, 3H), 7.07 (d, J = 8.7 Hz, 2H), 7.00-6.88 (m, 4H), 6.88-6.84 (m, 1H), 5.58 (s, 1H), 2.29 (s, 3H).
Lead Reference
- Tetramethoxybenzene is a Good Building Block for Molecular Wires: Insights from Photoinduced Electron Transfer
Used Chemicals
Procedure
To a 3-necked 300 mL round bottom flask was charged with diphenylamine (5.01 g, 29.6 mmol, 1.0 eq.), 4-chloroanisole (4.48 g, 31.4 mmol, 1.05 eq.) and degassed toluene (150 mL). To this solution was added Pd2(dba)3 (0.287 g, 0.131 mmol, 1 mol%), tri-tert-butylphosphonium tetrafluoroborate (0.198 g, 0.683 mmol, 2 mol%) and sodium tert-butoxide (6.34 g, 66.0 mmol, 2.2 eq.). The reaction mixture was refluxed for 16 hr under nitrogen atmosphere. After cooled to room temperature, the reaction was diluted with CH2Cl2 (300 mL). The suspension was filtered and the filtrate was dried over Na2SO4 and concentrated under reduced pressure to afford the crude and brown solid. The crude product was purified by silica-gel column chromatography (hexane/EtOAc = 99/1 then 8/1) to afford the light brown solid (7.0 g) containing 10 mol% of diphenylamine. Removal of the residual diphenylamine by recrystallization from hexane (55 mL, 60 °C then 15 °C) gave 4-methoxytriphenylamine as a white solid (5.26 g, 65 %).
Experimenter’s Comments
The reaction mixture was monitored by TLC (EtOAc/hexane = 1/10. Starting materials: Rf = 0.36 (diphenylamine), 0.59 (4-chloroanisole); target product: Rf = 0.46).
Analytical Data(4-Methoxytriphenylamine)
1H NMR (400 MHz, CD2Cl2); δ 7.26-7.17 (m, 4H), 7.10-6.98 (m, 6H), 6.98-6.91 (m, 2H), 6.89-6.82 (m, 2H), 3.79 (s, 3H).
13C NMR (101 MHz, CD2Cl2); δ 156.72, 148.56, 141.00, 129.39, 127.72, 123.12, 122.15, 115.05, 55.77.
Lead References
- Air-Stable Trialkylphosphonium Salts: Simple, Practical, and Versatile Replacements for Air-Sensitive Trialkylphosphines. Applications in Stoichiometric and Catalytic Processes
- An Air and Thermally Stable One- Component Catalyst for the Amination of Aryl Chlorides
Other References
- A Simple Catalytic Method for the Conversion of Aryl Bromides to Arylamines
- Palladium-catalyzed synthesis of arylamines from aryl halides. Mechanistic studies lead to coupling in the absence of tin reagents
- Air-stable trialkylphosphonium salts


References
[产品拾贝] Air-stable Tri-tert-butylphosphine Equivalent
[TCI应用实例] 使用Pd2(dba)3和tBu3P·HBF4的Buchwald-Hartwig 胺化反应
化学品安全说明书(SDS)
如需更多帮助,请联系我 们。
技术规格
CoA及其他文档
示例 CoA
目前没有该产品的 CoA 示例。
分析图谱

很抱歉,您搜索的分析图谱无法提供。







