Product Document Searching Made Easy by 2D Code! | TCI Materials Science News November 2025 | [TCIPracticalExample] Ullmann-type Coupling Reaction at Room Temperature... | Various analytical charts can be searched on each product detail page and Product Document Search (The kinds of analytical charts differ by product)
Maximum quantity allowed is 999
Please select the quantity
CAS RN: 170787-99-2 | Product Number: E1356
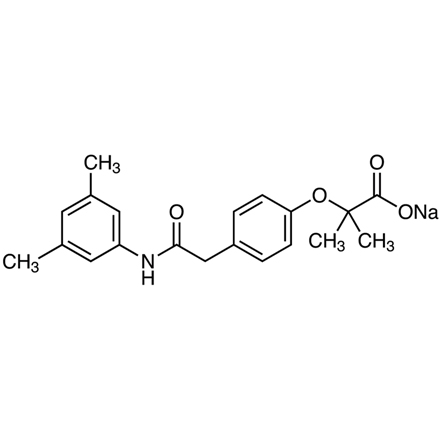
Efaproxiral Sodium
Purity: >98.0%(T)(HPLC)
Synonyms:
- RSR 13 Sodium
- Sodium 2-[4-[2-[(3,5-Dimethylphenyl)amino]-2-oxoethyl]phenoxy]-2-methylpropanoate
- 2-[4-[2-[(3,5-Dimethylphenyl)amino]-2-oxoethyl]phenoxy]-2-methylpropanoic Acid Sodium Salt
Product Documents:
| Size | Unit Price | Same Day | 2-3 Business Days | Other Lead Time |
Shipping Information

|
|---|---|---|---|---|---|
| 250MG |
$156.00
|
9 | 11 | Contact Us |
* Please contact our distributors or
TCI
to order our products. The above prices do not include freight cost, customs, and other charges to the destination.
* The storage conditions are subject to change without notice.
| Product Number | E1356 |
Purity / Analysis Method 
|
>98.0%(T)(HPLC) |
| Molecular Formula / Molecular Weight | C__2__0H__2__2NNaO__4 = 363.39 |
| Physical State (20 deg.C) | Solid |
Storage Temperature 
|
Frozen (-20°C) |
| Store Under Inert Gas | Store under inert gas |
| Condition to Avoid | Hygroscopic,Heat Sensitive |
| CAS RN | 170787-99-2 |
| Reaxys Registry Number | 8959239 |
| PubChem Substance ID | 468591726 |
| MDL Number | MFCD02093864 |
Specifications
| Appearance | White to Almost white powder to lump |
| Purity(HPLC) | min. 98.0 area% |
| Purity(Nonaqueous Titration) | min. 98.0 %(calcd.on anh.substance) |
| Melting point | 240.0 to 244.0 °C |
| Water | max. 13.0 % |
| NMR | confirm to structure |
Properties (reference)
GHS
Related Laws:
Transport Information:
| H.S.code* | 2924.29-000 |
Application
Efaproxiral Sodium: Developed as an Allosteric Modifier of Hemoglobin and a Radiation Sensitizer
Efaproxiral sodium (RSR 13 sodium), an analogue of the antilipemic agent bezafibrate [B3346], was developed as an allosteric modifier of hemoglobin1,2) and a radiation sensitizer.3) In the 2000s, efaproxiral sodium was tested in a clinical trial to increase the efficacy of certain chemotherapy agents which have reduced efficacy against hypoxic tumors, and can thus be made more effective by increased offloading of oxygen from hemoglobin-bound oxygen into the tumor tissues.4) But the clinical trial was dropped out in Phase III trials. In 2009, Efaproxiral was added to the World Anti-Doping Agency (WADA) list of Prohibited Substances as an artificially enhancer of the uptake, transport or delivery of oxygen.5) (The product is for research purpose only.)
References
- 1) Allosteric modifiers of hemoglobin. 1. Design, synthesis, testing, and structure-allosteric activity relationship of novel hemoglobin oxygen affinity decreasing agents
- 2) Allosteric modifiers of hemoglobin: 2-[4-[[(3,5-disubstituted anilino)carbonyl]methyl]phenoxy]-2-methylpropionic acid derivatives that lower the oxygen affinity of hemoglobin in red cell suspensions, in whole blood, and in vivo in rats
- 3) Radiation sensitization with redox modulators: a promising approach (a review)
- 4) Effects of RSR13 and oxygen on the cytotoxicity of cisplatin and carboplatin to EMT6 mouse mammary tumor cells in vitro and in vivo
- 5) Preventive doping control screening analysis of prohibited substances in human urine using rapid-resolution liquid chromatography/high-resolution time-of-flight mass spectrometry
Product Documents (Note: Some products will not have analytical charts available.)
Safety Data Sheet (SDS)
Please select Language.
The requested SDS is not available.
Please Contact Us for more information.
Specifications
C of A & Other Certificates
Please enter Lot Number
Incorrect Lot Number. Please input only the 4-5 alphanumeric characters before the hyphen.
Sample C of A
This is a sample C of A and may not represent a recently manufactured lot of the product.
A sample C of A for this product is not available at this time.
Analytical Charts

Please enter Lot Number
Incorrect Lot Number. Please input only the 4-5 alphanumeric characters before the hyphen.
The requested analytical chart is not available. Sorry for the inconvenience.



