Maintenance Notice (5:00 PM July 4 - 11:00 PM July 6, 2025):
This website is scheduled to be unavailable due to maintenance. We appreciate your patience and understanding.
Published TCIMAIL newest issue No.198
Maximum quantity allowed is 999
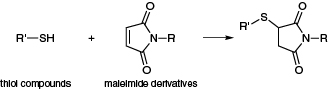
Figure 1. Reaction of thiol compounds and maleimides
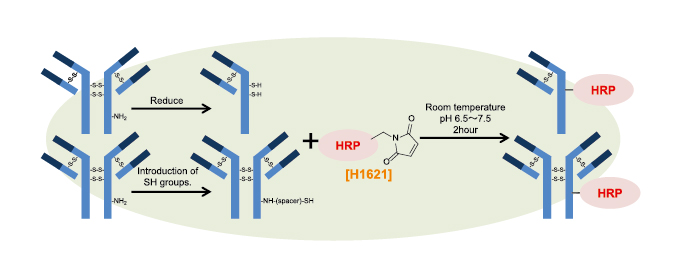
Figure 2. HRP-labelling of an antibody with 2