盲盒好礼,精彩继续 | 轻松扫码查看产品文档 | TCIMAIL No.199 已上新 | TCI试剂——品质可靠,值得信赖
订购方法?联系方式:021-67121386 / Sales-CN@TCIchemicals.com
Maximum quantity allowed is 999
请选择数量
CAS RN: 682-30-4 | 产品编码: D3728
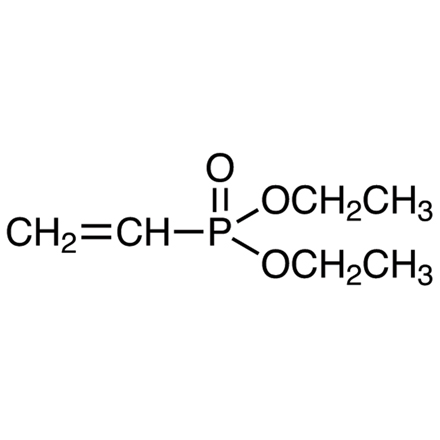
Diethyl Vinylphosphonate
* 点击“查询”可查看预计发货日期,仅供参考。
* 无具体发货日期的情况,如:显示“8个工作日后发货”,将在您订购日起的8个工作日后发货。
* 我们将以最优方式从上海/天津两大仓库发货。国内库存不足,需两周左右向日本总部调货。
* 对于可分装产品,11:30前的订单,当天发货;11:30后的订单,隔天发货。
* 如需大包装,请点击“大包装询价”按钮(对于某些产品我们无法提供大包装)。
* TCI会经常复审储藏条件以对其进行优化,请以在线目录为准,敬请留意。
* 更多信息,请联系营业部:021-67121386 / Sales-CN@TCIchemicals.com 。任何货期、规格或包装方面的需求,请联系我们 。
* 无具体发货日期的情况,如:显示“8个工作日后发货”,将在您订购日起的8个工作日后发货。
* 我们将以最优方式从上海/天津两大仓库发货。国内库存不足,需两周左右向日本总部调货。
* 对于可分装产品,11:30前的订单,当天发货;11:30后的订单,隔天发货。
* 如需大包装,请点击“大包装询价”按钮(对于某些产品我们无法提供大包装)。
* TCI会经常复审储藏条件以对其进行优化,请以在线目录为准,敬请留意。
* 更多信息,请联系营业部:021-67121386 / Sales-CN@TCIchemicals.com 。任何货期、规格或包装方面的需求,请联系我们 。
技术规格
| Appearance | Colorless to Almost colorless clear liquid |
| Purity(GC) | min. 98.0 % |
物性(参考值)
| 沸点 | 82 °C/15 mmHg |
| 比重 | 1.06 |
| 折射率 | 1.43 |
GHS
相关法规
| RTECS# | TA1150000 |
| 新化学物质备案回执号 | B1A232214944 |
运输信息
| 监管条件代码(*) |
应用
A Stereoselective synthesis of Z-Enamides

Experimental procedure1): to a dry flask is charged with amide (0.35 mmol), PdCl2(PhCN)2 (0.017 mmol), and CuCl (0.035 mmol). The reaction vessel is charged with oxygen followed by the addition of chlorobenzene (2 mL) and tetraethyl methylenediphosphonate (TEMDP, 0.035 mmol). The reaction mixture is stirred at 70 °C for 1 min and then diethyl vinylphosphonate (1.05 mmol) is added. The reaction mixture is stirred at 70 °C for 48 h under oxygen atmosphere. After the reaction is over, the solvent is evaporated under reduced pressure and the crude residue is purified by flash column chromatograph.
References
- 1) Hydrogen-Bond-Directed Highly Stereoselective Synthesis of Z-Enamides via Pd-Catalyzed Oxidative Amidation of Conjugated Olefins
- 2) Pd(II)-Catalyzed Hydroxyl-Directed C-H Olefination Enabled by Monoprotected Amino Acid Ligands
参考文献







