The relationship between genes and diseases has been studied extensively since the completion of human genome project in 2003. The direct cause of these diseases is sometimes related to the proteins produced in the human body by the human genome. The study of these proteins, "Proteomics" is very important in order to understand the relationship between genes and diseases.
The general method for protein analysis is isolation of the targeted protein by 2-D gel electrophoresis, followed by digestion with proteases to yield peptide fragment mixtures, which are then analyzed by MS/MS to identify the fragments, from which the isolated protein can then be reconstructed. However several problems still remain with 2-D gel electrophoresis, as extremely acidic, basic, or hydrophobic proteins cannot be fully separated. Furthermore, only the highly skilled experts are able to manage the 2-D gel electrophoresis to obtain reproducible data. For these reasons, new and improved methods for protein analysis have been explored.
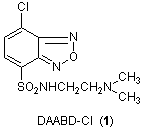
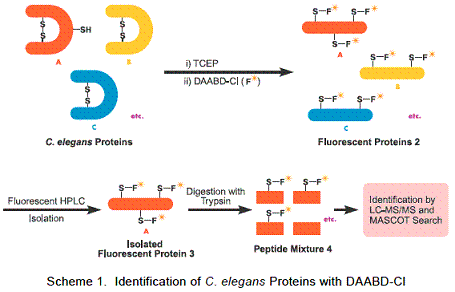
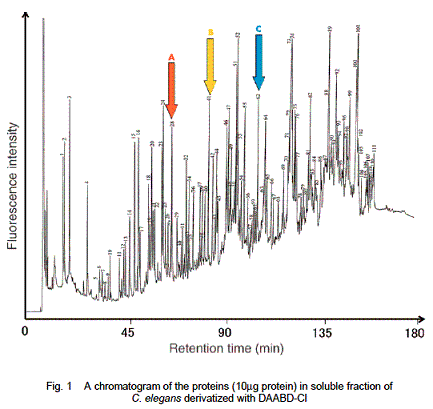
Imai and co-workers have developed a new method for protein analysis with use of DAABD-Cl (1). This new method can analyze proteins with high precision. Imai and co-workers extracted proteins from C. elegans, and the extracted proteins were first reacted with tris(2-carboxyethyl)phosphine in a buffer solution in order to reductively cleave the S-S bonds to yield the primary proteins. The resulting SH functional groups of resulting proteins were derivatized by reaction with DAABD-Cl to yield fluorescent labeled protein mixtures (2). The fluorescent labeled protein mixtures were separated by fluorescence HPLC to obtain fractions consisting of DAABD labeled proteins (Figure 1). The selected DAABD labeled protein (3) was isolated and digested using trypsin to obtain the peptide mixtures (4) consisting of DAABD labeled peptides and other peptides. The peptide mixtures were analyzed by LC-MS/MS and the resulting mass spectral data were analyzed to identify the original protein by the MASCOT database system (Scheme 1).
The chlorine at 7 position of DAABD-Cl reacts specifically with SH groups. DAABD-Cl itself is nonfluorescent, however the resultant DAABD-derivative is strongly fluorescent, due to the benzoxadiazole skeleton coupled to the SH group. Generally, there are not many S-S bonds and SH group in proteins, and consequently target proteins can be labeled with DAABD-Cl in an efficient manner. Additionally, both excitation and emission wavelengths of DAABD derivatives are long, allowing highly sensitive and selective protein analysis. Furthermore, DAABD-Cl has a dimethylamino group at 4 position, and therefore high intensity cations can be obtained with electron spray ionization during MS analysis. Therefore, extremely small quantities of peptides can be analyzed.
DAABD-Cl is a labeling reagent, which can effectively permit the collection of the target protein through fluorescence HPLC and analysis by MS/MS. This protein analysis reagent that Imai and co-workers have developed allows one to identify a very small amount of protein with good precision. It is expected that this technique can be used in many applications, including the identification of abnormal or pathogenic proteins in living organism.
References
- 1) M. Masuda, C. Toriumi, T. Santa, K. Imai, Anal. Chem. 2004, 76, 728.

- 2) M. Masuda, H. Saimaru, N. Takamura, K. Imai, Biomed. Chromatogr. 2005, 19, 556.

- 3) T. Ichibangase, K. Moriya, K. Koike, K. Imai, J. Proteome Res. 2007, 6, 2841.

- 4) H. Asamoto, T. Ichibangase, K. Uchikura, K. Imai, J. Chromatogr. A 2008, 1208, 147.

- 5) T. Ichibangase, H. Saimaru, et al., Biomed. Chromatogr. 2008, 22, 232.

- 6) K. Imai, T. Ichibangase, R. Saitoh, Y. Hoshikawa, Biomed. Chromatogr. 2008, 22, 1304.

- 7) T. Ichibangase, K. Imai, J. Proteome Res. 2009, 8, 2129.

- 8) K. Imai, A. Koshiyama, K. Nakata, Biomed. Chromatogr. 2011, 25, 59.

- 9) K. Nakata, R. Saitoh, J. Amano, A. Koshiyama, T. Ichibangase, et al., Cytokine 2012, 59, 317.

- 10) K. Imai, JP Patent 4558297.
- 11) Quantitative Proteome Analysis: Methods and Applications, ed. by K. Imai, S. L. F. Yau, Pan Stanford Publishing, Singapore, 2013.
- 12) K. Nakata, T. Ichibangase, R. Saitoh, M. Ishigai, K. Imai, Analyst 2015, 140, 71.

Page Top





![DAABD-Cl [=4-[2-(Dimethylamino)ethylaminosulfonyl]-7-chloro-2,1,3-benzoxadiazole] [for Proteome Analysis] DAABD-Cl [=4-[2-(Dimethylamino)ethylaminosulfonyl]-7-chloro-2,1,3-benzoxadiazole] [for Proteome Analysis]](/medias/A5596.jpg?context=bWFzdGVyfHJvb3R8Mzg0MTR8aW1hZ2UvanBlZ3xhRGcwTDJneFlTODRPVEk0TlRFNE9UTXdORFl5TDBFMU5UazJMbXB3Wnd8Njk3M2ZiZTE0NTgzMzNhMjFkZDE4MDU4ODJjYmZiM2IyYjg0NGYwOGI0ZGY2YjYzNjE0NGI2YjU4ZGU0Mzc2Mw)