Maintenance notice in CEST time zone (1:00 - 3:30 AM May 17, 2026): Our website is scheduled for maintenance. The website will be available, but some functions may experience errors. We appreciate your patience and understanding.
Published TCIMAIL newest issue No.201
Maximum quantity allowed is 999
Please select the quantity
CAS RN: 346440-54-8 | Product Number: B4138
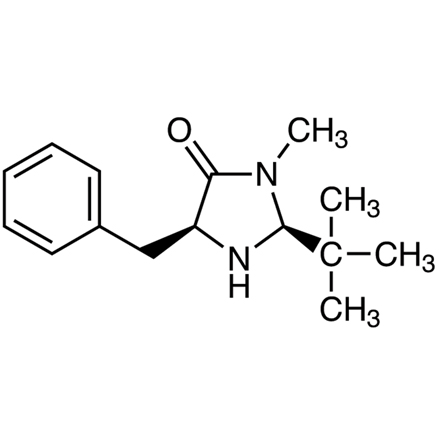
(2S,5S)-(-)-2-tert-Butyl-3-methyl-5-benzyl-4-imidazolidinone
Purity: >97.0%(GC)
Synonyms:
- (2S,5S)-(-)-5-Benzyl-2-tert-butyl-3-methyl-4-imidazolidinone
Product Documents:
* Stock available in Belgium: Shipment on the same day
* Stock available in Japan: Please check the Shipping Simulation for estimated shipments. (excludes regulated items and dry ice shipments)
| Product Number | B4138 |
Purity / Analysis Method 
|
>97.0%(GC) |
| Molecular Formula / Molecular Weight | C__1__5H__2__2N__2O = 246.35 |
| Physical State (20 deg.C) | Solid |
Storage Temperature 
|
Room Temperature (Recommended in a cool and dark place, <15°C) |
Packaging and Container 
|
200MG-Glass Bottle with Plastic Insert (View image) |
| CAS RN | 346440-54-8 |
| Reaxys Registry Number | 6480916 |
| PubChem Substance ID | 354334237 |
| MDL Number | MFCD03426982 |
Specifications
| Appearance | White to Light yellow powder to crystal |
| Purity(HPLC) | min. 98.0 area% |
| Optical purity(LC) | min. 98.0 ee% |
| Melting point | 97.0 to 101.0 °C |
Properties (reference)
| Melting Point | 100 °C |
| Specific Rotation | -60° (C=1,MeOH) |
GHS
Related Laws:
Transport Information:
| HS Number | 2933299090 |
Application
Imidazolidinone Derivatives for Versatile Asymmetric Reactions

References
- 1) The First Enantioselective Organocatalytic Mukaiyama-Michael Reaction: A Direct Method for the Synthesis of Enantioenriched γ-Butenolide Architecture
- 2) Enantioselective organocatalytic epoxidation using hypervalent iodine reagents
- 3) Enantioselective Organocatalytic Singly Occupied Molecular Orbital Activation: The Enantioselective α-Enolation of Aldehydes
- 4) Enantioselective Organocatalytic Intramolecular Diels−Alder Reactions. The Asymmetric Synthesis of Solanapyrone D
- 5) Total Synthesis of (−)-Spinosyn A via Carbonylative Macrolactonization
PubMed Literature
Product Articles
[Product Highlights] Imidazolidinone Derivatives for Versatile Asymmetric ReactionsProduct Documents (Note: Some products will not have analytical charts available.)
Safety Data Sheet (SDS)
Please select Language.
The requested SDS is not available.
Please Contact Us for more information.
Specifications
C of A & Other Certificates
Please enter Lot Number
Incorrect Lot Number. Please input only the 4-5 alphanumeric characters before the hyphen.
Sample C of A
This is a sample C of A and may not represent a recently manufactured lot of the product.
A sample C of A for this product is not available at this time.
Analytical Charts

Please enter Lot Number
Incorrect Lot Number. Please input only the 4-5 alphanumeric characters before the hyphen.
The requested analytical chart is not available. Sorry for the inconvenience.