Maximum quantity allowed is 999
Please select the quantity
CAS RN: 851030-18-7 | Product Number: B4587
2,4-Bis(benzyloxy)-6-chloro-1,3,5-triazine

Purity: >95.0%(T)(HPLC)
Synonyms:
- DBT-Cl
- CDBT
Product Documents:
* Available Stock: Prompt shipment (for products) in Saitama/Hyogo warehouse. Stock In Other WH: Shipment in about 2-3 Business Days from Saitama warehouse. Please contact us
if you need further information.
* The displayed price is the unit price and does not include consumption tax. The unit prices displayed are the latest and are subject to change without notice.
* To send your quote request for bulk quantities, please click on the "Request Bulk Quote" button. Please note that we cannot offer bulk quantities for some products.
*TCI frequently reviews storage conditions to optimize them. Please note that the latest information on the storage temperature for the products is described on our website.
* The displayed price is the unit price and does not include consumption tax. The unit prices displayed are the latest and are subject to change without notice.
* To send your quote request for bulk quantities, please click on the "Request Bulk Quote" button. Please note that we cannot offer bulk quantities for some products.
*TCI frequently reviews storage conditions to optimize them. Please note that the latest information on the storage temperature for the products is described on our website.
| Product Number | B4587 |
Purity / Analysis Method 
|
>95.0%(T)(HPLC) |
| Molecular Formula / Molecular Weight | C__1__7H__1__4ClN__3O__2 = 327.77 |
| Physical State (20 deg.C) | Solid |
Storage Temperature 
|
Room Temperature (Recommended in a cool and dark place, <15°C) |
Packaging and Container 
|
1G-Glass Bottle with Plastic Insert (View image) |
| CAS RN | 851030-18-7 |
| Reaxys Registry Number | 15462339 |
| PubChem Substance ID | 354333516 |
Specifications
| Appearance | White to Almost white powder to crystal |
| Purity(HPLC) | min. 95.0 area% |
| Purity(Argentometric Titration) | min. 95.0 % |
| Melting point | 64.0 to 68.0 °C |
| NMR | confirm to structure |
Properties (reference)
| Melting Point | 66 °C |
GHS
| Pictogram |

|
| Signal Word | Warning |
| Hazard Statements | H315 : Causes skin irritation. H319 : Causes serious eye irritation. |
| Precautionary Statements | P264 : Wash skin thoroughly after handling. P280 : Wear protective gloves/ eye protection/ face protection. P302 + P352 : IF ON SKIN: Wash with plenty of water. P337 + P313 : If eye irritation persists: Get medical advice/ attention. P305 + P351 + P338 : IF IN EYES: Rinse cautiously with water for several minutes. Remove contact lenses, if present and easy to do. Continue rinsing. P362 + P364 : Take off contaminated clothing and wash it before reuse. P332 + P313 : If skin irritation occurs: Get medical advice/ attention. |
Related Laws:
Transport Information:
Application
A Triazine Derivative Usable for the Glycosylation Reaction of Unprotected Sugars
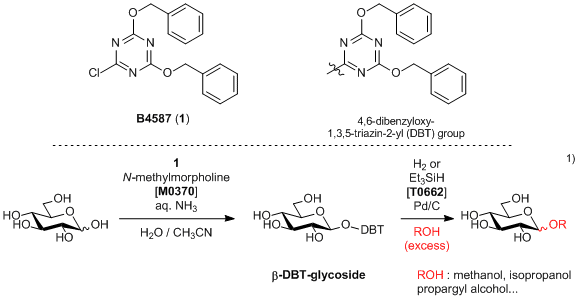
Experimental procedure1): 2,4-bis(benzyloxy)-6-chloro-1,3,5-triazine (1, 12.3 g, 37.5 mmol) is added to a solution of D-glucose (4.5 g, 25 mmol), N-methylmorpholine (4.1 mL, 38 mmol) and ammonia (3.3 mL, 50 mmol, 29% aqueous solution) in the mixture of acetonitrile and water (125 mL, CH3CN:H2O = 1:1) and the resulting mixture is stirred for 3 h at 0 °C. After concentration in vacuo, the residue is washed with water and chloroform. The filtrate is concentrated in vacuo to give β-glucoside (9.42 g, 20.0 mmol, 80 % yield).
Excess amount of alcohol is added to the mixture of β-glucoside (0.2 mmol) and Pd/C (5 mg). Under hydrogen atmosphere, the resulting mixture is stirred at room temperature. The reaction mixture is filtered, and the filtrate is concentrated in vacuo to afford the alkoxy group substituted glucosides.
Excess amount of alcohol is added to the mixture of β-glucoside (0.2 mmol) and Pd/C (5 mg). Under hydrogen atmosphere, the resulting mixture is stirred at room temperature. The reaction mixture is filtered, and the filtrate is concentrated in vacuo to afford the alkoxy group substituted glucosides.
References
- 1)Protection-free Synthesis of Alkyl Glycosides under Hydrogenolytic Conditions
PubMed Literature
Product Articles
[Product Highlights] A Triazine Derivative Usable for the Glycosylation Reaction of Unprotected SugarsProduct Documents (Note: Some products will not have analytical charts available.)
Safety Data Sheet (SDS)
Please select Language.
The requested SDS is not available.
Please Contact Us for more information.
Specifications
C of A & Other Certificates
Please enter Lot Number
Incorrect Lot Number. Please input only the 4-5 alphanumeric characters before the hyphen.
Sample C of A
This is a sample C of A and may not represent a recently manufactured lot of the product.
A sample C of A for this product is not available at this time.
Analytical Charts

Please enter Lot Number
Incorrect Lot Number. Please input only the 4-5 alphanumeric characters before the hyphen.
The requested analytical chart is not available. Sorry for the inconvenience.





