Published TCIMAIL newest issue No.200
Maximum quantity allowed is 999
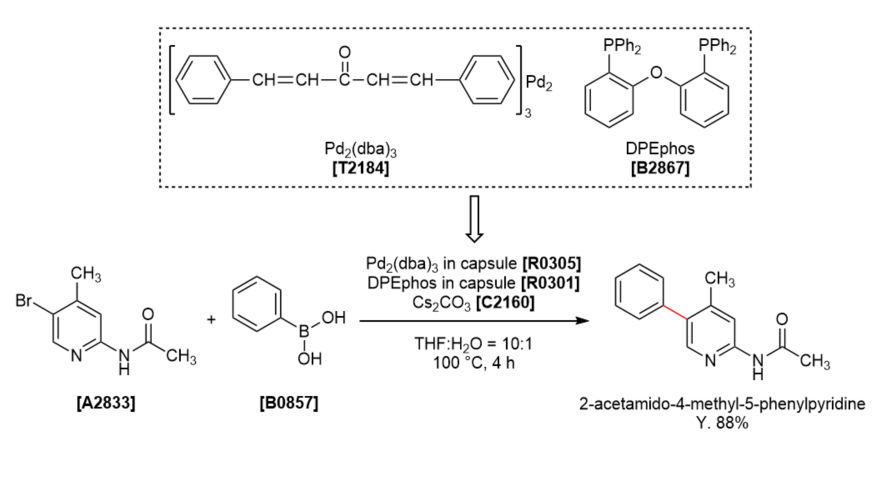
TCI Practical Example: Suzuki-Miyaura Coupling Using Encapsulated Pd2(dba)3 and Encapsulated DPEphos
We are proud to present the Suzuki-Miyaura coupling of 2-acetamido-5-bromo-4-methylpyridine and phenylboronic acid using the encapsulated Pd2(dba)3 and the encapsulated DPEphos. Capsule reagents can omit weighing the catalyst and ligand. Each capsule includes ca. 60 μmol of Pd2(dba)3 and ca. 150 μmol of DPEphos, respectively.
Used Chemicals
Procedure
Pd2(dba)3 (HPMC encapsulated) (one capsule) and DPEphos (HPMC encapsulated) (one capsule) were added to a solution of 2-acetamido-5-bromo-4-methylpyridine (340 mg, 1.50 mmol), cesium carbonate (540 mg, 2.0 mmol, 1.1 eq.) and phenylboronic acid (270 mg, 2.3 mmol, 1.5 eq.) in THF/water (10:1, 8.25 mL) at room temperature and the reaction mixture was stirred at 100 °C for 4 hours. The solution was then diluted with ethyl acetate (10 mL) and sat. sodium sulfate (10 mL), and filtered through Celite pad. The filtrate was separated and the organic layer was washed with water (15 mL) and brine (15 mL), dried over sodium sulfate and concentrated under reduced pressure. The crude product was purified by silica gel chromatography (ethyl acetate:hexane = 1:2 - 1:1) to afford 2-acetamido-4-methyl-5-phenylpyridine (319 mg, 94% yield) as a white solid.
Experimenter's Comments
The reaction mixture was monitored by UPLC/MS.
The material of the capsule is water-soluble HPMC (hydroxypropyl methylcellulose).
The shape of the capsule is like the image below and Pd2(dba)3 (ca. 60 µmol) and DPEphos (ca. 150 µmol) are encapsulated in each capsule, respectively.
The capsule disintegrates as shown in the image below when stirred in the solvent for about 15 minutes, releasing its content.

Analytical Data
2-Acetamido-4-methyl-5-phenylpyridine
1H NMR (400 MHz, CDCl3); δ 9.24–9.06 (brs, 1H), 8.16 (s, 1H), 8.10 (s, 1H), 7.46–7.36 (m, 3H), 7.30–7.29 (m, 2H), 2.32 (s, 3H), 2.21 (s, 3H).
Lead Reference
- Reagent and Catalyst Capsules: A Chemical Delivery System for Reaction Screening and Parallel Synthesis
Other References
- HPMC: A Biomass-Based Semisynthetic Sustainable Additive Enabling Clean and Fast Chemistry in Water

