Product Document Searching Made Easy by 2D Code! | TCI Chemistry News November 2025 | [TCIPracticalExample] The Z-Selective Horner-Wadsworth-Emmons Reaction... | Various analytical charts can be searched on each product detail page and Product Document Search (The kinds of analytical charts differ by product)
Maximum quantity allowed is 999
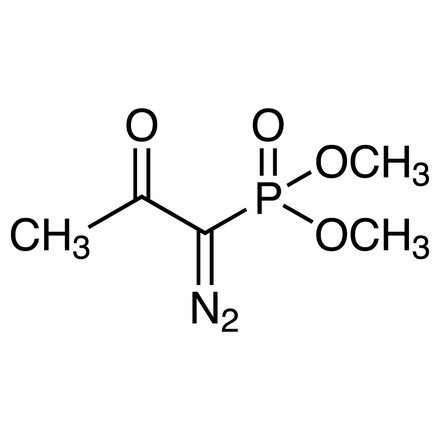
CAS RN: 90965-06-3 | 產品號碼: D3546
Dimethyl (1-Diazo-2-oxopropyl)phosphonate
* 以上價格已含運費關稅等但一些需要海運以及乾冰運輸的產品除外,詳情請與
當地經銷商
洽詢。
* TCI會時常優化儲存條件,儲存溫度請以在線目錄為準,敬請留意。
| Appearance | Light yellow to Brown clear liquid |
| Purity(HPLC) | min. 97.0 area% |
| 比重 | 1.28 |
| 折射率 | 1.48 |
| HS編碼* | 2931.49-900 |

-
Used Chemicals
-
Procedure
-
To a methanol (10 mL) solution of undecanal (0.20 g, 1.2 mmol) was added potassium carbonate (0.32 g,2.4 mmol) and Ohira-Bestmann reagent (0.27 g, 1.4 mmol) at room temperature. The reaction mixture was stirred overnight at room temperature, then diluted with diethyl ether and washed with saturated aqueous sodium bicarbonate, and dried by sodium sulfate. The organic layer was concentrated under reduced pressure. The resulting crude product was purified by column chromatography (hexane:toluene = 3:1 on silica gel) to give 1 as a colorless liquid (0.11 g, 56% yield).
-
Experimenter’s Comments
-
The reaction mixtures were monitored by 1H NMR (CDCl3).
-
Analytical Data(1-Dodecyne (1))
-
1H NMR (400 MHz, CDCl3); δ 2.18 (t, 2H, J = 7.1 Hz), 1.94 (s, 1H), 1.57-1.46 (m, 2H), 1.44-1.33 (m, 2H), 1.33-1.19 (m, 12H), 0.88 (t, 3H, J = 6.4 Hz).
13C NMR (101 MHz, CDCl3); δ 85.0, 68.2, 32.0, 29.7, 29.7, 29.5, 29.3, 28.9, 28.6, 22.8, 18.5, 14.3.
-
Lead Reference
-
- Further Improvements of the Synthesis of Alkynes from Aldehydes
-
Other References
-
- Methanolysis of Dimethyl (1-Diazo-2-oxopropyl) Phosphonate: Generation of Dimethyl (Diazomethyl) Phosphonate and Reaction with Carbonyl Compounds

References

References
- S. Ohira, Synth. Commun. 1989, 19, 561.

- S. Muller, B. Liepold, G. J. Roth, H. J. Bestmann, Synlett 1996, 521.

- H. D. Dickson, S. C. Smith, K. W. Hinkle, Tetrahedron Lett. 2004, 45, 5597.

- E. Quesada, R. J. K. Taylor, Tetrahedron Lett. 2005, 46, 6473.

文檔
[TCI Practical Example] Construction of the Terminal Alkyne Using Ohira-Bestmann Reagent
SDS
請求的SDS不可用。
如需更多幫助,請聯繫我們 。
產品規格
檢驗報告(CoA)及其他文檔
示例 CoA
目前沒有該產品的 CoA 示例。
分析圖譜

很抱歉,您搜索的分析圖譜無法提供。





