Published TCIMAIL newest issue No.201
Maximum quantity allowed is 999
*Lagerbestand in Belgien: Versand am selben Tag
*Lagerbestand in Japan: Bitte prüfen Sie die Versandsimulation für voraussichtliche Lieferzeiten. (ausgenommen regulierte Produkte und Trockeneissendungen)
| Artikel # | B3960 |
Reinheit / Analysenmethode 
|
>94.0%(T) |
| Summenformel / Molekülmasse | C__6H__1__2N__2O__4S__2 = 240.29 |
| Physikalischer Zustand (20 °C) | Solid |
Lagerungstemperatur 
|
Refrigerated (0-10°C) |
| Unter Inertgas lagern | Store under inert gas |
| Zu vermeidende Bedingungen | Air Sensitive,Heat Sensitive |
Verpackung und Behälter 
|
1G-Glass Bottle with Plastic Insert (Bild ansehen) |
| CAS RN | 119752-83-9 |
| Reaxys Registrierungsnummer | 21034099 |
| PubChem-Stoff-ID | 354335121 |
| Appearance | White to Almost white powder to crystal |
| Purity(Iodometric back titration) | min. 94.0 % |
| Schmelzpunkt | 180 °C(dec.) |
| Löslichkeit in Wasser | Soluble |
| Piktogramm |

|
| Signalwort | Achtung |
| Gefahrenhinweise | H315 : Verursacht Hautreizungen. H319 : Verursacht schwere Augenreizung. |
| Sicherheitshinweise | P264 : Nach Gebrauch Haut gründlich waschen. P280 : Schutzhandschuhe/ Augenschutz/ Gesichtsschutz tragen. P302 + P352 : BEI BERÜHRUNG MIT DER HAUT: Mit viel Wasser waschen. P337 + P313 : Bei anhaltender Augenreizung: Ärztlichen Rat einholen/ ärztliche Hilfe hinzuziehen. P362 + P364 : Kontaminierte Kleidung ausziehen und vor erneutem Tragen waschen. P332 + P313 : Bei Hautreizung: Ärztlichen Rat einholen/ ärztliche Hilfe hinzuziehen. |
| HS-Nr. (Import / Export) (TCI-E) | 2933599590 |

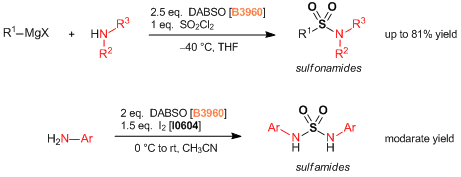
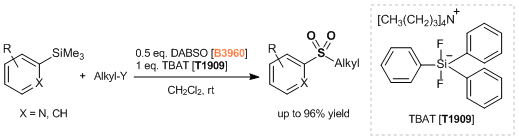
Under nitrogen atmosphere, a degassed DMI (20 mL) solution of 4-biphenylboronic acid (792 mg, 4.00 mmol), DABSO (577 mg, 2.40 mmol), tmphen (95 mg, 0.40 mmol), lithium tert-butoxide (320 mg, 4.00 mmol) and NiBr2(glyme) (123 mg, 0.40 mmol) was stirred at 100 °C for 14 hours. The mixture was cooled to room temperature and tert-butyl bromoacetate (1.17 mL, 8.00 mmol) was added and the reaction mixture stirred for 1 hour. Water (20 mL) was poured into the reaction mixture and the mixture was extracted with diethyl ether (50 mL x 3). The combined organic phase was washed with water (50 mL) and brine (40 mL), dried over sodium sulfate and filtered. The solvent was removed under reduced pressure and the residue was purified by medium pressure preparative chromatography (ethyl acetate:hexane = 5:95 - 15:85 on silica gel) to give compound 1 as a white solid (1.01 g, 76%).
NiBr2(glyme) and LiOtBu were weighed into a sample tube in a glove box because these reagents are moisture sensitive. If these were used in air, it should be weighted quickly and used up the reagents as soon as possible after opening.
Since the ligand of NiBr2(glyme) may be desorbed under reduced pressure, depressurization operations is desirable to be finished as soon as possible.
The reaction mixture was monitored by TLC (ethyl acetate:hexane = 2:1, Rf = 0.38) and UPLC-MS.
Compound 1
1H NMR (400 MHz, CDCl3); δ 8.00 (d, 2H, J = 8.9 Hz), 7.78 (m, 2H), 7.58–7.66 (m, 2H), 7.42-7.54 (m, 3H), 4.08 (s, 2H), 1.58 (s, 2H), 1.39 (s, 9H).

To a solution of 4-bromobiphenyl (1.17 g, 5.00 mmol), DABSO (721 mg, 3.00 mmol), and PdCl2(Amphos)2 (177 mg, 0.25 mmol) in anhydrous isopropyl alcohol (20 mL) was added triethylamine (2.1 mL, 15 mmol) at room temperature and the mixture was stirred at 75 °C for 23 hours. The suspension was cooled at room temperature and NFSI (2.36 g, 7.5 mmol) was added and the reaction mixture stirred for 3 hours until completion. The solvent was removed under reduced pressure. The residue was diluted with ethyl acetate (20 mL) and filtrated through celite pad. The filtrate was washed with saturated aqueous Na2S2O3 solution (20 mL) and brine (20 mL), dried over sodium sulfate and filtered. The solvent was removed under reduced pressure and the residue was purified by medium pressure preparative chromatography (ethyl acetate:hexane = 2:98 - 5:95 on silica gel) to give compound 1 as a white solid (928 mg, 79%).
The reaction mixture was monitored by TLC (ethyl acetate:hexane = 1:9, Rf = 0.57) and UPLC-MS.
Compound 1
1H NMR (400 MHz, CDCl3); δ 8.12–8.04 (m, 2H), 7.86–7.80 (m, 2H), 7.67–7.58 (m, 2H), 7.44-7.55 (m, 3H).

Das angeforderte SDB ist nicht verfügbar.
Bitte Kontaktieren Sie uns für mehr Informationen.
Ein Muster-AZ für dieses Produkt ist zur Zeit nicht verfügbar.

Das angeforderte Analysediagramm ist nicht verfügbar. Wir entschuldigen uns für die Unannehmlichkeiten.