It has come to our notice that certain fraudulent individuals or entities are misusing our Company’s name and TCI’s registered trademarks by promoting and offering regulated and hazardous chemical substances through online platforms like YouTube. We hereby categorically clarify that TCI has no association or connection whatsoever with the products being displayed or sold in the videos. These products have been falsely represented as being associated with TCI, and the unauthorized use of our trademark and brand name is both illegal and misleading. TCI Chemicals markets and sells its products exclusively through its official website and authorized distributors. If you become aware of any such fraudulent activity or require clarification, you may reach out to us at: Sales-IN@TCIchemicals.com. Click Here to View the Caution Notice.
Product Document Searching Made Easy by 2D Code! | [Product Highlights] Endogenous Biotin-Blocking Reagent...Maximum quantity allowed is 999
CAS RN: 14338-32-0 | Product Number: C0903
2-Chloro-1-methylpyridinium Iodide
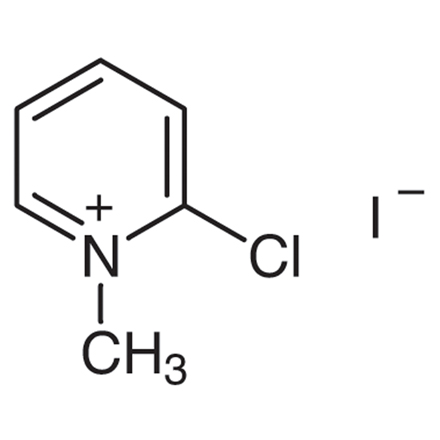
Purity: >98.0%(T)
| Size | Unit Price | Hyderabad | Japan* | Quantity |
|---|---|---|---|---|
| 25G |
₹4,300.00
|
9 | 38 |
|
*Upon orders receipt, Hyderabad stocks will be dispatched on the same day.
*Items available in Japan warehouse will be dispatched in 10-12 working days.
*INR price is exclusive of domestic taxes applicable.
*TCI frequently reviews storage conditions to optimize them. Please note that the latest information on the storage temperature for the products is described on our website.
| Product Number | C0903 |
| Purity / Analysis Method | >98.0%(T) |
| Molecular Formula / Molecular Weight | C__6H__7ClIN = 255.48 |
| Physical State (20 deg.C) | Solid |
| Storage Temperature | Room Temperature (Recommended in a cool and dark place, <15°C) |
| Store Under Inert Gas | Store under inert gas |
| Condition to Avoid | Light Sensitive,Hygroscopic |
| CAS RN | 14338-32-0 |
| Reaxys Registry Number | 3572320 |
| PubChem Substance ID | 87565959 |
| SDBS (AIST Spectral DB) | 10163 |
| Merck Index (14) | 6301 |
| MDL Number | MFCD00011984 |
| Appearance | Light yellow to Amber to Dark green powder to crystal |
| Purity(HPLC) | min. 95.0 area% |
| Purity(Nonaqueous Titration) | min. 98.0 % |
| Solubility in Water | almost transparency |
| Pictogram |

|
| Signal Word | Warning |
| Hazard Statements | H315 : Causes skin irritation. H319 : Causes serious eye irritation. |
| Precautionary Statements | P264 : Wash skin thoroughly after handling. P280 : Wear protective gloves/ eye protection/ face protection. P302 + P352 : IF ON SKIN: Wash with plenty of water. P337 + P313 : If eye irritation persists: Get medical advice/ attention. P305 + P351 + P338 : IF IN EYES: Rinse cautiously with water for several minutes. Remove contact lenses, if present and easy to do. Continue rinsing. P362 + P364 : Take off contaminated clothing and wash it before reuse. P332 + P313 : If skin irritation occurs: Get medical advice/ attention. |

-
Used Chemicals
-
Procedure
-
Boc-Pro-OH (1.65 g, 7.65 mmol), 2-chloro-1-methylpyridinium iodide (1.95 g, 7.65 mmol) were added to solution of H-Phe-OMe・HCl (1.50 g,6.95 mmol) in dichloromethane (30 mL) at room temperature and triethylamine (3.40 mL, 24.3 mmol) was added dropwise in 3 min at 5 to 10 °C. After stirring for 3 h, solvent was removed under reduced pressure and the residue was dissolved in ethyl acetate and washed with 10% citric acid aq. (30 mL × 3), 5% sodium bicarbonate solution aq. (30 mL × 3) and brine (30 mL). The organic layer was dried over anhydrous sodium sulfate and filtered. After the removal of solvent under reduced pressure, the given crude was purified by column chromatography (hexane : EtOAc = 2 : 1) to give Boc-Pro-Phe-OMe (2.37 g,91%) as a slightly yellowish solid.
-
Experimenter's Comments
-
Consumption of stating material and formation of product was monitored by LCMS.
-
Analytical Data(Boc-Pro-Phe-OMe)
-
1H NMR (400 MHz, DMSO-d6); δ 8.17–8.27 (m, 1H), 7.19–7.29 (m, 5H), 4.42–4.53 (m, 1H), 4.00–4.13 (m, 1H), 3.56–3.61 (m, 3H), 3.18–3.38 (m, 2H), 2.90–3.08 (m, 2H), 1.91–2.11 (m, 1H), 1.56–1.71 (m, 3H), 1.15-1.40 (m, 9H).
-
Lead Reference
-
- A facile synthesis of carboxamides by using 1-methyl-2-halopyridinium iodides as coupling reagents

References
- 1) A CONVENIENT METHOD FOR THE SYNTHESIS OF CARBOXYLIC ESTERS
- 2) A FACILE SYNTHESIS OF CARBOXAMIDES BY USING 1-METHYL-2-HALOPYRIDINIUM IODIDES AS COUPLING REAGENTS
- 3) Asymmetric Decarboxylative Allylation of Oxindoles
Safety Data Sheet (SDS)
The requested SDS is not available.
Please Contact Us for more information.
Specifications
C of A & Other Certificates
Sample C of A
A sample C of A for this product is not available at this time.
Analytical Charts
The requested analytical chart is not available. Sorry for the inconvenience.





