Maximum quantity allowed is 999
CAS RN: 185990-03-8 | Product Number: D4357
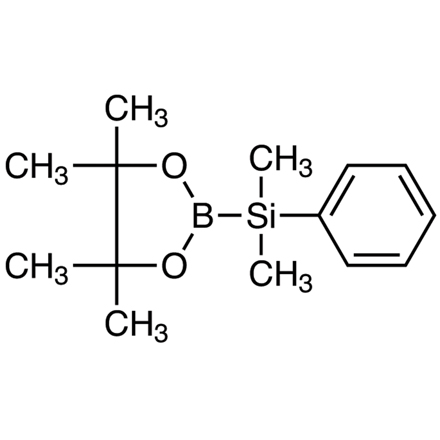
2-(Dimethylphenylsilyl)-4,4,5,5-tetramethyl-1,3,2-dioxaborolane
Purity: >95.0%(GC)
- (Dimethylphenylsilyl)boronic Acid Pinacol Ester
| Size | Unit Price | Hyderabad | Japan* | Quantity |
|---|---|---|---|---|
| 1G |
₹8,200.00
|
7 | 14 |
|
| 5G |
₹19,800.00
|
2 | ≥60 |
|
*Upon orders receipt, Hyderabad stocks will be dispatched on the same day.
*Items available in Japan warehouse will be dispatched in 10-12 working days.
*INR price is exclusive of domestic taxes applicable.
*TCI frequently reviews storage conditions to optimize them. Please note that the latest information on the storage temperature for the products is described on our website.
| Product Number | D4357 |
Purity / Analysis Method 
|
>95.0%(GC) |
| Molecular Formula / Molecular Weight | C__1__4H__2__3BO__2Si = 262.23 |
| Physical State (20 deg.C) | Liquid |
Storage Temperature 
|
Room Temperature (Recommended in a cool and dark place, <15°C) |
| Store Under Inert Gas | Store under inert gas |
| Condition to Avoid | Air Sensitive,Moisture Sensitive |
| CAS RN | 185990-03-8 |
| Reaxys Registry Number | 7585086 |
| PubChem Substance ID | 253661961 |
| MDL Number | MFCD05664111 |
| Appearance | Colorless to Light yellow to Light orange clear liquid |
| Purity(GC) | min. 95.0 % |
| Boiling Point | 120 °C/0.08 mmHg |
| Specific Gravity (20/20) | 0.97 |
| Refractive Index | 1.50 |
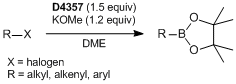
To a vial sealed with a screw cap containing a silicon-coated rubber septum is added potassium methoxide (0.6 mmol) under argon. It is connected to a vacuum/nitrogen manifold through a needle. DME (5 mL) and 1 (0.36 mmol) are added to the vial, then stirred for 10 min at 30 °C. 4-Bromoanisole (0.50 mmol) is added dropwise and stirred for 1 h. The solution is cooled to 0 °C followed by the addition of TBAF (1.0 M, 800 µL). The resultant solution is stirred for 3 h at 0 °C. After that, the mixture is added to the H2O (100 mL), then extracted with Et2O (50 mL). The organic layer is washed with water (50 mL x 2). The combined organic layer is then dried over MgSO4 followed by evaporation. The crude product is purified by boric acid-impregnated silica-gel column chromatography with 0–5% hexane/Et2O eluent to give the desired product (77% isolated yield) as a colorless oil.
References
- Anomalous Reactivity of Silylborane: Transition-Metal-Free Boryl Substitution of Aryl, Alkenyl, and Alkyl Halides with Silylborane/Alkoxy Base Systems
Documents
Safety Data Sheet (SDS)
The requested SDS is not available.
Please Contact Us for more information.
Specifications
C of A & Other Certificates
Sample C of A
A sample C of A for this product is not available at this time.
Analytical Charts

The requested analytical chart is not available. Sorry for the inconvenience.





