Sign up for free ground shipping on all website orders, no minimum required, and get exclusive coupons!
Maximum quantity allowed is 999
CAS RN: 3240-34-4 | Product Number: I0330
Iodobenzene Diacetate
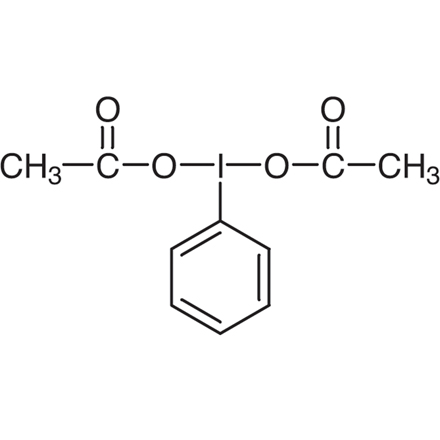
Purity: >97.0%(T)
- (Diacetoxyiodo)benzene
- PIDA
- DIB
| Size | Unit Price | Philadelphia, PA | Portland, OR | Japan* | Quantity |
|---|---|---|---|---|---|
| 10G |
$27.00
|
2 | 1 | ≥100 |
|
| 25G |
$52.00
|
4 | 3 | ≥100 |
|
| 250G |
$329.00
|
Contact Us | 1 | 31 |
|
* Items in stock locally ship in 1-2 business days. Items from Japan stock are able to ship from a US warehouse within 2 weeks. Please contact TCI for lead times on items not in stock. Excludes regulated items and items that ship on ice.
* To send your quote request for bulk quantities, please click on the "Request Quote" button. Please note that we cannot offer bulk quantities for some products.
*TCI frequently reviews storage conditions to optimize them. Please note that the latest information on the storage temperature for the products is described on our website.
| Product Number | I0330 |
Purity / Analysis Method 
|
>97.0%(T) |
| Molecular Formula / Molecular Weight | C__1__0H__1__1IO__4 = 322.10 |
| Physical State (20 deg.C) | Solid |
Storage Temperature 
|
Room Temperature (Recommended in a cool and dark place, <15°C) |
| Store Under Inert Gas | Store under inert gas |
| Condition to Avoid | Moisture Sensitive |
| CAS RN | 3240-34-4 |
| Reaxys Registry Number | 1879369 |
| PubChem Substance ID | 87571686 |
| SDBS (AIST Spectral DB) | 12662 |
| MDL Number | MFCD00008692 |
| Appearance | White to Light yellow powder to crystal |
| Purity(Iodometric Titration) | min. 97.0 % |
| Solubility in Methanol | almost transparency |
| Melting Point | 158 °C(dec.) |
| Solubility in water | Insoluble |
| Solubility (soluble in) | Methanol |
| Solubility (insoluble in) | Ether |
| Pictogram |

|
| Signal Word | Warning |
| Hazard Statements | H315 : Causes skin irritation. H319 : Causes serious eye irritation. |
| Precautionary Statements | P264 : Wash skin thoroughly after handling. P280 : Wear protective gloves/ eye protection/ face protection. P337 + P313 : If eye irritation persists: Get medical advice/ attention. P305 + P351 + P338 : IF IN EYES: Rinse cautiously with water for several minutes. Remove contact lenses, if present and easy to do. Continue rinsing. P302 + P352 : IF ON SKIN: Wash with plenty of soap and water. P332 + P313 : If skin irritation occurs: Get medical advice/ attention. P362 : Take off contaminated clothing and wash before reuse. |
| RTECS# | DA3525000 |
| HS Number | 2915.39.3500 |

-
Used Chemicals
-
Procedure
-
To a solution of 1-naphthalenemethanol (306 mg, 2.0 mmol) in dichloromethane (2 mL, 1.0 mol/L) was added TEMPO (31.2 mg, 0.20 mmol), PhI(OAc)2 (709mg, 2.2 mmol) and the mixture was stirred at room temperature for 4 hours. Dichloromethane (12.5 mL), saturated aqueous sodium thiosulfate solution (12.5 mL) was added and the mixture was stirred for 30 minutes. The organic layer was washed with saturated aqueous sodium bicarbonate solution (10 mL), brine (10 mL) and the organic layer was dried over sodium sulfate and filtered. The solvent was removed under reduced pressure and the residue was purified by column chromatography (ethyl acetate:hexane = 0:100 - 3:97 on silica gel) to give 1-naphthaldehyde as a yellow liquid (291 mg, 93%).
-
Experimenter’s Comments
-
The reaction mixture was monitored by TLC (ethyl acetate:hexane = 1:9, Rf = 0.50).
-
Analytical Data
-
1-Naphthaldehyde
1H NMR (400 MHz, CDCl3); δ 10.41 (s, 1H), 9.26 (d, J = 8.9 Hz, 1H), 8.11 (d, J = 8.4 Hz, 1H), 8.01 (d, J = 6.8 Hz, 1H), 7.93 (d, J = 8.9 Hz, 1H), 7.74-7.57 (m, 3H).
-
Lead Reference
-
- 2-(Hydroxyimino)aldehydes: Photo- and Physicochemical Properties of a Versatile Functional Group for Monomer Design

-
Used Chemicals
-
Procedure
-
To a solution of 2-phenylimidazoline (500 mg, 3.4 mmol), potassium carbonate (520 mg, 3.8 mmol, 1.1 equiv) in DMSO (17 mL) was added iodobenzene diacetate (1.21 g, 3.8 mmol, 1.1 equiv) and stirred at rt for 18 hours. The residue was diluted with ethyl acetate (75 mL) and washed with saturated aqueous sodium bicarbonate solution (50 mL, twice) dried over sodium sulfate and filtered. The solvent was removed under reduced pressure and the residue was purified by column chromatography (on aminosilica gel, ethyl acetate:hexane = 0:1 - 1:0), giving 2-phenylimidazole as a white solid (340 mg,y. 69%).
-
Experimenter’s Comments
-
The reaction mixture was monitored by NH-TLC (ethyl acetate, Rf = 0.30).
-
Analytical Data
-
2-phenylimidazole
1H NMR (270 MHz, DMSO-d6); δ 12.5 (dr, 1H), 7.95-7.91 (m, 2H), 7.44 (t, 2H, J = 7.3 Hz), 7.33 (t, 1H, J = 7.3 Hz), 7.24 (s, 1H), 7.02 (s, 1H)
-
Lead Reference
-
- Expanded applicability of iridium(I) NHC/phosphine catalysts in hydrogen isotope exchange processes with pharmaceutically-relevant heterocycles

References
- G. Piancatelli, F. Leonelli, Org. Synth. 2006, 83, 18.
Documents
[Research Articles] Synthesis of 1,2-Diamine Derivatives Using a Rhodium Catalyst
Safety Data Sheet (SDS)
The requested SDS is not available.
Please Contact Us for more information.
Specifications
C of A & Other Certificates
Sample C of A
A sample C of A for this product is not available at this time.
Analytical Charts

The requested analytical chart is not available. Sorry for the inconvenience.





